POSTERS AND PUBLICATIONS
Previous ASM Clinical Virology Symposium (CVS) Posters/Presentations
Development of Synthetic Multiplexed External Controls for Monitoring the Performance of Qualitative Laboratory Nucleic Acid Testing Panels Used for Rapid Identification of Blood Culture Pathogens.

This poster was presented at the May 2019 Clinical Virology Symposium, Savannah GA, (USA). Maine Molecular Quality Controls Inc. (MMQCI) has developed unique, extractable multiplex control panels designed to monitor all pathogenic organisms and antibiotic resistance genes detected by GenMark’s ePlex® Blood Culture Identification (BCID) Gram-positive (GP), Gram-negative (GN), and Fungal Pathogen (FP) Panels.
Validation of a multiplexed synthetic control panel to monitor the performance of simultaneous detection of multiple respiratory viral, bacterial pathogens, and antimicrobial resistance genes in lower respiratory infections.
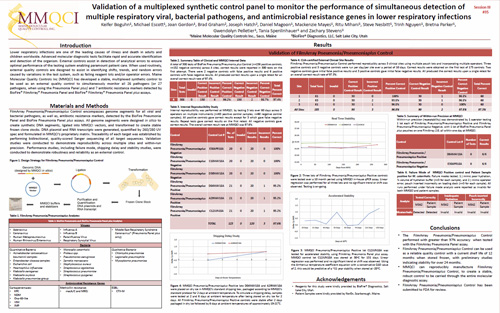
This poster was presented at the May 2019 Clinical Virology Symposium, Savannah GA, (USA).Maine Molecular Quality Controls Inc has validated and received FDA clearance for a stable, multiplexed synthetic control to be used as an external quality control to simultaneously monitor all 26 pathogens (or 27 pathogens, when using the Pneumonia Panel plus) and 7 antibiotic resistance markers detected by BioFire® FilmArray® Pneumonia and Pneumonia plus Panel.
Development of Synthetic Multiplexed External Controls for Monitoring the Performance of a Qualitative Laboratory Nucleic Acid Testing Panel Used for Rapid Identification of Respiratory Pathogens.
