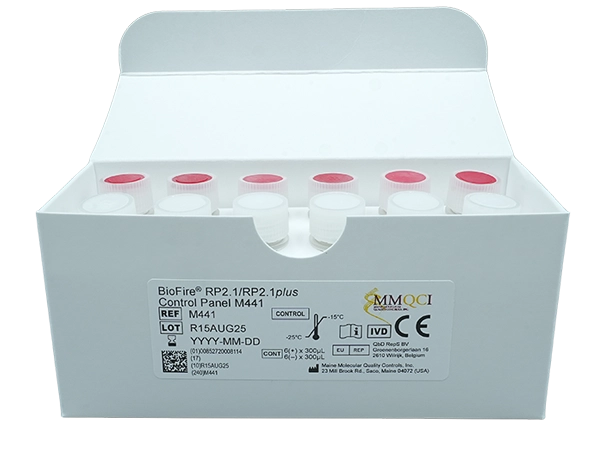
BioFire® RP2.1/RP2.1plus Control Panel M441
The BioFire RP2.1/RP2.1plus Control Panel M441 is intended for in vitro use as reference material to monitor the detection and identification of respiratory viral and bacterial targets in nasopharyngeal swabs (NPS) obtained from individuals suspected of respiratory tract infections.
Product Information:
BioFire® RP2.1/RP2.1plus Control Panel M441
Part Number: M441
GTIN: 00852720008114
Kit Contains: 12 tubes x 300µL
Controls/Qty: 6 x Positive M44321, 6 x Negative M44221
Storage: Frozen (–25°C to -15°C)