eplex BCID-GN Control M326
The eplex BCID-GN Control M326 is intended for in vitro use as a qualitative external quality control to monitor the amplification, detection and identification steps on the cobas® eplex blood culture identification gram-negative (BCID-GN) panel performed on the cobas eplex system (Roche Diagnostics).
Product Information:
eplex BCID-GN Control M326
Part Number: M326
GTIN: Not available at this time
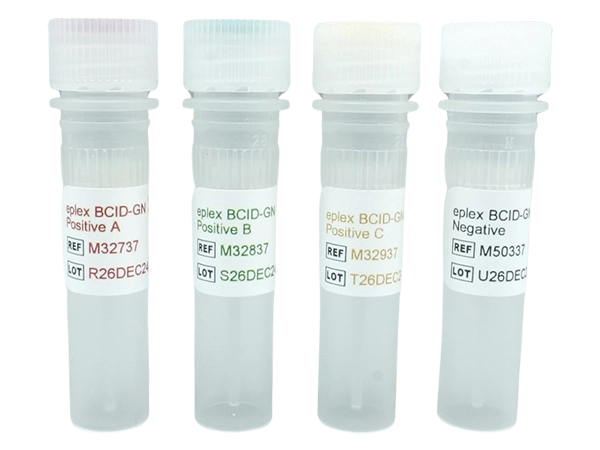
Kit Contains: 12 tubes x 50μL
Controls/Qty: 3 x Positive A M32737, 3 x Positive B M32837 , 3 x Positive C M32937, and 3 x Negative M50337
Storage: Refrigerated (2° – 8°C)
Documents:
You might be interested in: